Nurosym week 3: early HRV gains, but symptoms unchanged
Three weeks into taVNS, my HRV has risen 23% and hit a six-month high. My symptoms haven't shifted. This post covers the numbers, the confounders, and what I think they mean so far.
Three weeks into using the Nurosym taVNS device, my heart rate variability is showing the clearest sustained improvement I’ve recorded since becoming ill. But my day-to-day symptoms haven’t shifted. This post covers the numbers, the confounders, and what I think they mean so far.
The headline numbers
In the week before I started the Nurosym (14–20 March), my 7-day average HRV was 16.7ms. In the most recent week (4–10 April), it’s 20.6ms. That’s roughly a 23% increase, and my reading on 10 April (24ms) is the highest single value in my entire dataset going back to October 2025.
My resting heart rate has also dropped, from the low 80s in February and early March to a consistent 77–79 over the past two weeks.
For context, my HRV spent the five months from October to mid-March stuck in a narrow band between about 14 and 17. Small daily fluctuations, but no sustained upward movement. The shift over the past three weeks is qualitatively different from anything in my prior data.
The confounders
I’d love to be able to attribute this cleanly to one thing. I can’t. Three interventions overlap with this period:
Nurosym taVNS. I started daily 1-hour right tragus stimulation on 21 March, following the protocol described in my initial Nurosym post. The first visible uptick in my 7-day rolling HRV average appears within the first week of use.
Candesartan dose increase. My dose was increased from 4mg to 8mg on 25 March. Candesartan is an angiotensin receptor blocker, sometimes used in dysautonomia for its effects on blood pressure and potentially on autonomic regulation. The drop in my resting heart rate from the low 80s to 77–79 is consistent with the candesartan doing real work, and lower resting heart rate could itself support higher HRV readings. The evidence on candesartan is covered in the amitriptyline, propranolol, and candesartan review.
Amitriptyline taper. I reduced from 50mg to 37.5mg on 29 March, then to 25mg on 4 April. Amitriptyline has anticholinergic effects that suppress vagal tone, so reducing it could plausibly allow HRV to rise. However, I’d previously reduced from 100mg to 75mg (1 December) and from 75mg to 50mg (15 December) with no corresponding HRV improvement, which makes the amitriptyline taper a less convincing explanation for the current change.
I also started a graded recumbent exercise bike protocol on 23 February (starting at 10 minutes, three times weekly, progressing slowly). This predates the HRV improvement by nearly a month, so its contribution is harder to pin down. It’s possible the exercise is having a delayed effect, or that it’s providing a foundation that the other interventions are building on. The post-exertional malaise caveat applies here: I’ve been careful to stay well within any threshold that triggers worsening.
I’m aware that running multiple interventions simultaneously makes it impossible to isolate which one is driving the improvement. In an ideal world, I’d trial each change in isolation and wait months between them. In practice, I don’t have that luxury. I need to throw every potentially helpful, low-risk intervention at my health, and if benefits are realised, the lack of cleanly separable attribution is the least of my worries. This tension is part of why I try to track consistently — see what I’m tracking and why for the full picture.
What hasn’t changed: symptoms
My daily fatigue/breathlessness score has been a 7 out of 10 almost every day since late January. Head pain has been a steady 3 since mid-February. Neither has budged during the period of HRV improvement.
This isn’t unusual. Autonomic metrics like HRV and resting heart rate reflect changes in nervous system regulation that can precede symptomatic improvement by weeks or longer. The Stavrakis trial measured outcomes at 2 months, and I’m only at 3 weeks. But it’s worth being honest about: the numbers on the screen are improving, and I don’t feel any different.
For a broader picture of what the diaphragmatic breathing work has shown — another intervention targeting vagal tone through a different mechanism — the pattern of metric improvement ahead of symptom improvement appears there too.
What the chart shows
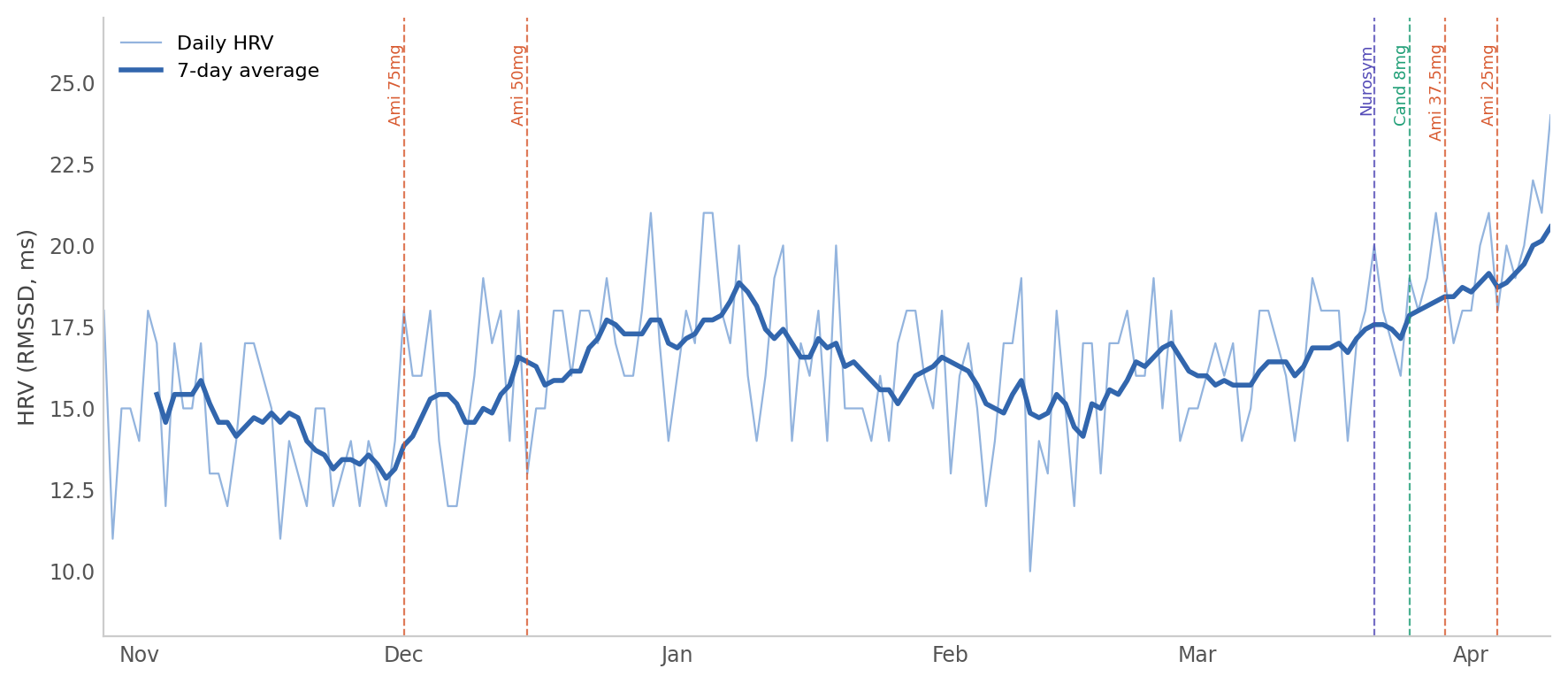
The chart plots daily HRV readings (thin line) and a 7-day rolling average (thick line) from 28 October 2025 to 10 April 2026. Vertical markers indicate the start of the Nurosym (21 March), the candesartan increase (25 March), and the two amitriptyline reductions (29 March and 4 April).
The rolling average is flat at around 15–16 for most of the dataset, begins to lift in the last week of March, and climbs more steeply in early April. The daily values remain noisy, but the recent cluster of readings in the 18–24 range is visibly different from the 12–18 band that characterised the preceding months.
Summary of period averages:
| Period | HRV average | RHR average |
|---|---|---|
| Oct–Nov 2025 (baseline) | 14.8 | 83 |
| Feb 2026 (pre-intervention) | 15.5 | 81 |
| 21–28 Mar (Nurosym week 1) | 18.5 | 80 |
| 4–10 Apr (most recent week) | 20.6 | 78 |
Early interpretation
If I had to weigh the likely contributors, the Nurosym looks like the strongest candidate for the initial step change. The first week of HRV improvement coincides with starting the device and precedes both the amitriptyline taper and the main candesartan effect on resting heart rate. The further acceleration in the most recent week could reflect the amitriptyline reduction, a cumulative Nurosym effect building over three weeks, or both.
I’m not drawing conclusions yet. Three weeks is too short, the confounders are real, and the symptom scores are a sobering reminder that autonomic metrics aren’t the whole picture. The next meaningful checkpoint is at 2 months (mid-May), matching the Stavrakis trial duration. I’ll report back then.
Previous post: Starting Nurosym: why I’m trying vagal nerve stimulation and what I’ll be looking for
The Nurosym device was purchased at my own expense. I have no affiliation with the manufacturer.